The Science Behind Dry Cleaning: Which Solvents Make It Work?
Dry cleaning may seem like a simple service, but behind the scenes it relies on chemistry, physics, and carefully chosen solvents to clean clothes without water. Unlike traditional washing, dry cleaning targets oil-based stains and delicate fabrics using specialized liquids that protect garment structure and appearance. This article explores the science behind dry cleaning, explaining which solvents make it work and why they are so effective.
What Makes Dry Cleaning Different from Washing?
Dry cleaning differs from regular laundry because it does not use water as the primary cleaning agent. Water causes fibers to swell, dyes to bleed, and some fabrics to shrink or lose shape. Dry cleaning avoids these problems by using chemical solvents that interact differently with fabric fibers.
The Role of Polarity in Cleaning
One of the key scientific principles behind dry cleaning is polarity. Water is a polar molecule, which means it attracts other polar substances like salts and sugars. Many stains, however, are non-polar, such as grease, oils, and waxes. Non-polar solvents used in dry cleaning dissolve these stains far more effectively than water.
The Science Behind Dry Cleaning: Which Solvents Make It Work?
Why Solvents Are Essential in Dry Cleaning
Solvents are liquids capable of dissolving other substances. In dry cleaning, solvents penetrate fabric fibers and loosen dirt without causing damage.
Protecting Fabric Structure
Natural fibers like wool and silk are especially sensitive to water. Solvents do not cause fibers to swell, helping garments maintain their original size and shape.
Efficient Stain Removal
Body oils, cosmetics, and food grease bond strongly to fabric. Dry cleaning solvents break these bonds at a molecular level, lifting stains away during the cleaning cycle.
The Most Common Dry Cleaning Solvents Explained
Perchloroethylene (Perc)
Perchloroethylene, commonly known as perc, has long been the most widely used dry cleaning solvent.
How Perc Works
Perc is a non-polar solvent that dissolves oils and grease quickly. Its low boiling point allows it to evaporate easily, leaving clothes dry at the end of the process.
Scientific Advantages
Perc is chemically stable, reusable through filtration, and highly effective across many fabric types. These properties made it the industry standard for decades.
Health and Environmental Considerations
While effective, perc can pose health risks with prolonged exposure and may contaminate soil or groundwater if improperly handled. This has driven research into alternative solvents.
Hydrocarbon Solvents
Hydrocarbon solvents are petroleum-derived liquids similar to mineral spirits.
The Chemistry Behind Hydrocarbon Solvents
These solvents are less aggressive than perc, meaning they interact more gently with dyes and fibers. Their molecular structure allows them to dissolve oils while reducing fabric stress.
Performance Trade-Offs
Hydrocarbon solvents may require longer cleaning cycles and higher temperatures to achieve the same results as perc, affecting energy efficiency.
Silicone-Based Solvents (D5)
Silicone-based solvents, especially decamethylcyclopentasiloxane (D5), represent a newer approach to dry cleaning.
Why Silicone Solvents Work
D5 molecules are large and smooth, allowing them to glide over fabric fibers while carrying away oils. This makes them especially suitable for delicate garments.
Environmental Breakdown
D5 breaks down into silica, carbon dioxide, and water under environmental conditions, which is why it is often marketed as a greener alternative.
Liquid Carbon Dioxide (CO₂)
Liquid CO₂ dry cleaning uses pressurized carbon dioxide instead of traditional chemical solvents.
The Physics of CO₂ Cleaning
Carbon dioxide becomes a liquid under high pressure. In this state, it behaves like a solvent, penetrating fabrics and dissolving oils. Once pressure is released, it returns to a gas, leaving no residue.
Advantages and Limitations
CO₂ is non-toxic, non-flammable, and environmentally friendly. However, the machinery required is expensive, limiting widespread adoption.
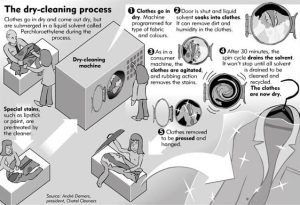
The Dry Cleaning Process Step by Step
Inspection and Pretreatment
Garments are inspected for stains, which may be pretreated using targeted spotting agents that work alongside the main solvent.
Solvent Cleaning Cycle
Clothes are placed in a machine similar to a washing machine. Solvent circulates through the fabrics, dissolving oils and lifting dirt.
Filtration and Reuse
Modern machines filter and distill solvents so they can be reused multiple times, reducing waste and cost.
Drying and Finishing
Heat or vacuum systems remove remaining solvent. Garments are then pressed, steamed, and shaped for a polished finish.
How Science Protects Colors and Fabrics
Minimal Dye Interaction
Dry cleaning solvents are designed to avoid dissolving fabric dyes, preventing color fading and bleeding.
Reduced Mechanical Stress
Because garments do not absorb solvent the way they absorb water, there is less agitation and friction during cleaning.
Eco-Friendly Innovations in Dry Cleaning Science
Professional Wet Cleaning
Wet cleaning uses controlled amounts of water and biodegradable detergents, combined with precise temperature and agitation control.
Bio-Based Solvents
Researchers are developing solvents derived from renewable resources that offer effective cleaning with lower environmental impact.
Safety Standards and Regulations
Worker Safety
Ventilation systems, sealed machines, and protective equipment reduce solvent exposure for dry cleaning workers.
Consumer Safety
Garments are aerated thoroughly to remove solvent residue, making professionally cleaned clothing safe to wear.
The Future of Dry Cleaning Solvents
The science behind dry cleaning continues to evolve. As environmental awareness grows and regulations tighten, the industry is shifting toward safer, more sustainable solvent systems. Advances in chemistry and engineering are making it possible to balance cleaning power with environmental responsibility.
Conclusion
The effectiveness of dry cleaning lies in science. By using carefully selected solvents, dry cleaners remove stains that water cannot while preserving delicate fabrics. From traditional perchloroethylene to innovative solutions like liquid CO₂ and silicone-based solvents, each option relies on chemical principles that make dry cleaning possible. Understanding the science behind these solvents helps consumers appreciate how dry cleaning works and why it remains an essential service for garment care.
